 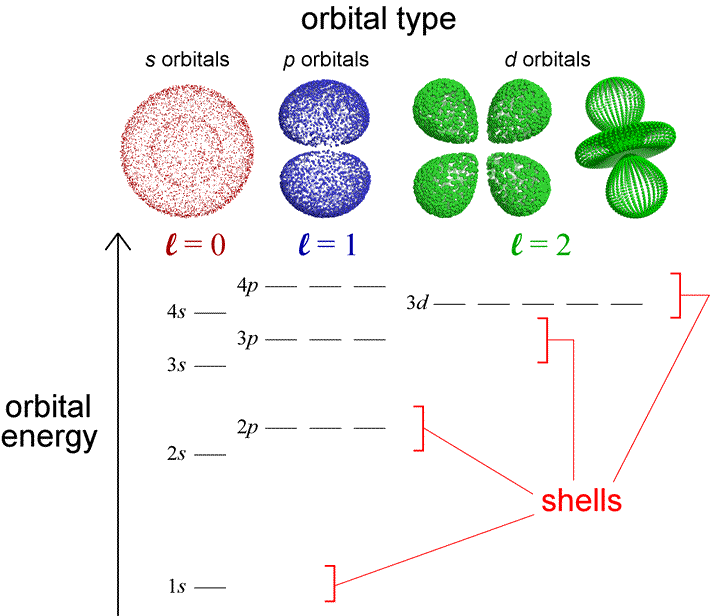
Electrons in the \(σ^∗_s\) orbitals are located well away from the region between the two nuclei. Just as with atomic orbitals, we create an energy-level diagram by listing the molecular orbitals in order of increasing energy. Adding electrons to these orbitals creates a force that holds the two nuclei together, so we call these orbitals bonding orbitals. The key difference is that in molecular orbitals, the electrons are allowed to interact with more than one atomic nucleus at a time. Electrons in a σ s orbital are attracted by both nuclei at the same time and are more stable (of lower energy) than they would be in the isolated atoms. The asterisk signifies that the orbital is an antibonding orbital. The out-of-phase addition (which can also be thought of as subtracting the wave functions) produces a higher energy \(σ^∗_s\) m olecular orbital (read as "sigma-s-star") molecular orbital in which there is a node between the nuclei. The in-phase combination produces a lower energy σ s molecular orbital (read as "sigma-s") in which most of the electron density is directly between the nuclei. The two types are illustrated in Figure 8.4.3. There are two types of molecular orbitals that can form from the overlap of two atomic s orbitals on adjacent atoms. (b) When out-of-phase waves combine, destructive interference produces a wave with less (or no) amplitude. creates bonds from overlap of atomic orbitals ( s, p, d ) and hybrid orbitals ( sp, sp2, sp3 ) combines atomic orbitals to form molecular orbitals (,, , ) forms or bonds. considers electrons delocalized throughout the entire molecule. 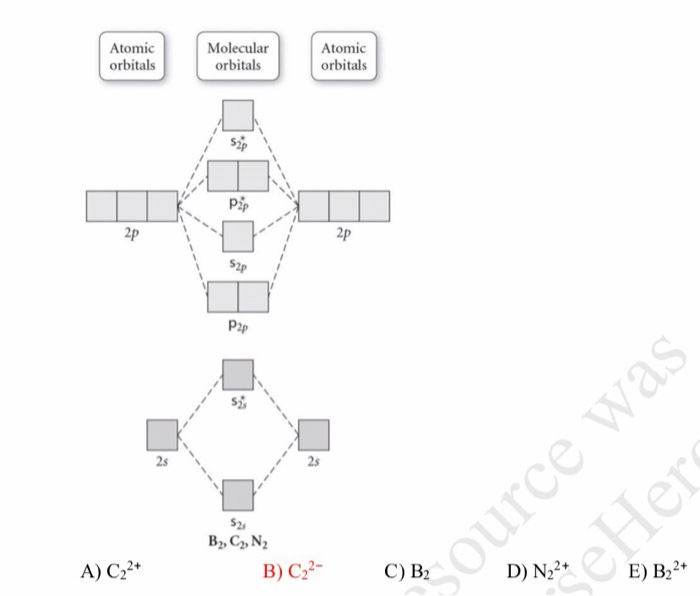
\): (a) When in-phase waves combine, constructive interference produces a wave with greater amplitude. considers bonds as localized between one pair of atoms. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
